 A strong absorbance band in the 1650-1750 cm -1 region indicate that a carbonyl group (C=O) is present. 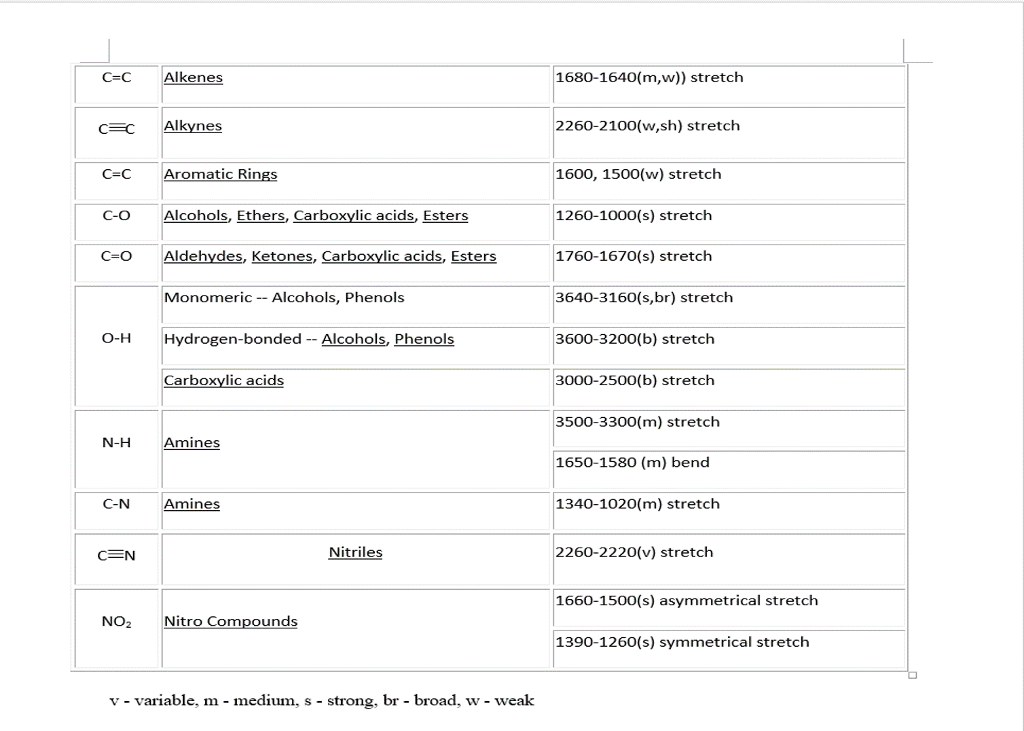
It is a very strong band comparing to the others on the spectrum. The technique is therefore very useful as a means of identifying which functional groups are present in a molecule of interest.įor example, the most characteristics absorption band in the spectrum of 2-hexanone ( Figure 6.3a) is that from the stretching vibration of carbonyl double bond C=O, at 1716 cm -1. The power of infrared spectroscopy arises from the observation that the covalent bonds characterizing different functional groups have different characteristic absorption frequencies (in wavenumber, Table 6.1 ). The larger wavenumbers (shorter wavelengths) are associated with higher frequencies and higher energy. Please note the direction of the horizontal axis (wavenumber) in IR spectra decrease from left to right. The wavenumber is defined as the reciprocal of wavelength ( Formula 6.3), and the wavenumbers of infrared radiation are normally in the range of 4000 cm -1 to 600 cm -1 (approximate corresponds the wavelength range of 2.5 μm to 17 μm of IR radiation). But instead of using frequency to show the absorbed radiation, wavenumbers (, in unit of cm -1) are used as a conventional way in IR spectra. The horizontal axis indicates the position of an absorption band.A molecule have a variety of covalent bonds, and each bond have different vibration modes, so the IR spectrum of a compound usually show multiple absorption bands. The spikes are called absorption bands in an IR spectrum. Lower values of % transmittance mean that some of the energy is absorbed by the compound, and gives the downward spikes. At the high end of the axis, 100% transmittance means no absorption occurred at that frequency. 
The solid line traces the values of % transmittance for every wavelength passed through the sample.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
